Janux Therapeutics, Inc. (NASDAQ:JANX) is a clinical-stage biopharmaceutical company that generates novel immunotherapies to modulate T cells to fight cancer. The company’s platforms engineer new drug candidates that overcome the limitations of current T-cell Engagers [TCEs] for solid tumor treatments. Today, Janux’s pipeline includes JANX007, which targets Prostate-Specific Membrane Antigen [PSMA] for metastatic castration-resistant prostate cancer [mCRPC], and JANX008, which targets Epidermal Growth Factor Receptor [EGFR] for several solid tumors. Therefore, I consider JANX a “buy” for investors who are aware of the inherent biotech and M&A risks.
TRACTr: Business Overview
Janux Therapeutics was founded in 2025 and is headquartered in San Diego, California. JANX’s proprietary Tumor-Activated T Cell Engager [TRACTr] and Tumor-Activated Immunomodulator [TRACIr] platforms engineer drug candidates designed to overcome the limitations of existing T-cell tumor treatments. The TRACTr platform designs molecules that selectively activate T cells only in the tumor microenvironment. At the same time, the TRACIr system generates molecules targeting tumor antigens to enhance immune response with reduced side effects.
Source: Corporate Presentation. May 2024.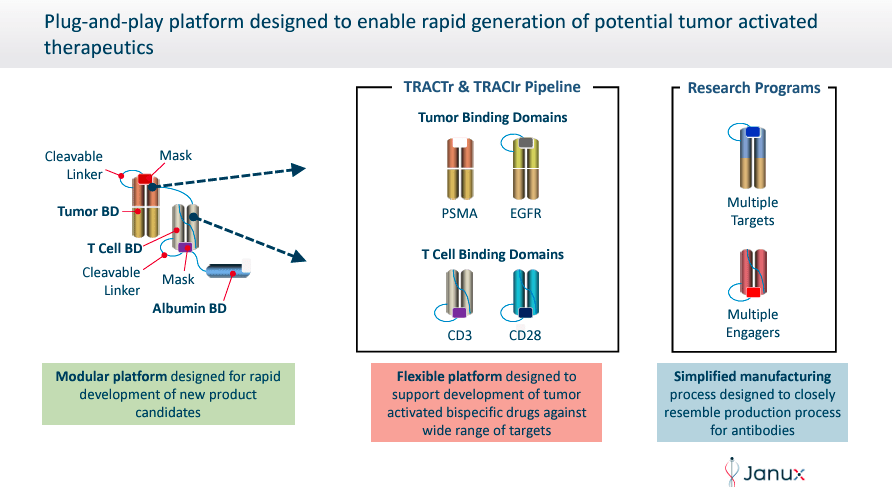
Moreover, the company’s TRACTr platform selectively creates activated molecules in the presence of tumor-specific proteases in the cancer microenvironment and stays inactive in healthy tissues. The TRACIr platform creates tumor-activated immunomodulators that use a tumor antigen-binding domain bonded to a T cell costimulatory CD28 binding domain. This design ensures that JANX’s action mechanism targets specifically cancerous cells, enhancing the immune response while reducing side effects.
Source: Janux Therapeutics’ website.
JANX’s pipeline includes two programs: JANX007 and JANX008. JANX007 is a bispecific molecule studied in Phase 1 clinical trials. This drug is the company’s leading tumor-activated T-cell engager [TRACTr], which targets Prostate-Specific Membrane Antigen [PSMA]. It is indicated for metastatic castration-resistant prostate cancer [mCRPC].
On the other hand, JANX008 is also a TRACTr targeting the Epidermal Growth Factor Receptor (EGFR) in Phase 1. However, JANX008 is indicated for multiple solid tumors, such as colorectal cancer [CRC], squamous cell carcinoma of the head and neck [SCCHN], non-small cell lung cancer [NSCLC], and renal cell carcinoma [RCC].
Cancer Treatment Results Propel Janux Therapeutics
More recently, on February 26, 2024, JANX announced positive safety and efficacy results for JANX007 in heavily pretreated patients with advanced mCRPC. The results suggest that JANX007 could stand as the best treatment for mCRPC due to its efficacy and safety profile. In patients receiving an initial dose of 0.2mg or higher, 83% (5 out of 6) achieved a 50% reduction in Prostate-Specific Antigen [PSA] levels.
This indicator, known as [PSA50], is a well-known biomarker for monitoring the disease because high PSA levels are related to the presence of prostate cancer. Additionally, another result showed that, with a first dose of 0.1mg or higher, 56% of patients (10 out of 18) reached a PSA50 reduction. The safety profile showed no Cytokine Release Syndrome [CRS] greater than grade 2 was observed. Therefore, there was no exaggerated immune response that could be harmful. This corroborates that JANX007 had a satisfactory safety profile and paved the way to phase 2 and 3 trials. It also shows that its effects are dose-dependent, often suggesting that the drug’s action mechanism works.
Additionally, JANX noted its early JANX008 data results presented differentiated safety and promising efficacy in heavily pretreated patients with various solid tumors. A subject with NSCLC, an aggressive lung cancer difficult to treat, achieved a Response Evaluation Criteria in Solid Tumors [RECIST] partial response [PR] maintained during 18 weeks with a 100% primary lung tumor reduction (i.e., the tumor disappeared).
Source: Corporate Presentation. May 2024.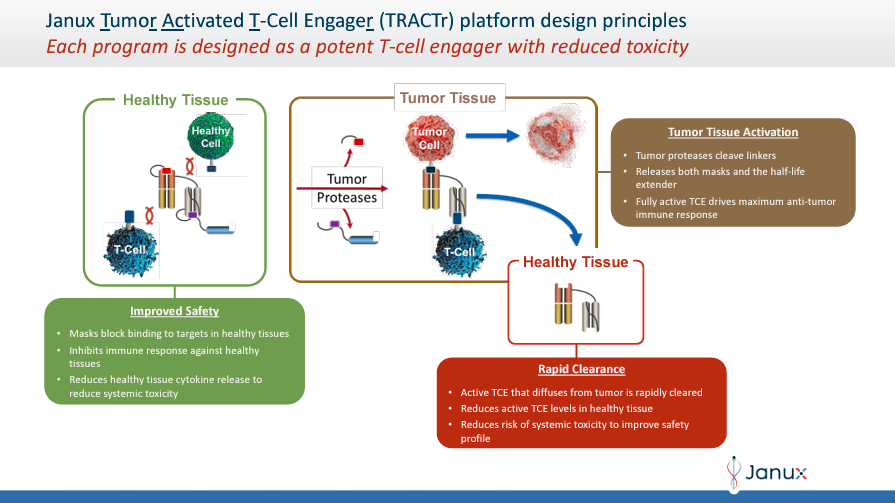
In fact, JANX’s drug managed to eliminate the liver’s metastasis without severe adverse events [SAEs] or dose-limiting toxicities [DLTs] were present. Furthermore, JANX’s executives mentioned they expect an update on JANX007 data in 2H2024. As for JANX008, we should get another update by 2025. The company is also conducting additional pre-clinical TRACTr and TRACIr programs for future studies. In my view, this is quite amazing, and it’s likely why the company’s valuation is now in the billions, as the medicine was effective and well-tolerated. So overall, I’d argue, its pipeline is advancing quite promisingly for effective cancer therapies.
Worth the Premium: Valuation Analysis
From a valuation perspective, JANX trades at a $2.1 billion market cap. This is significant as the company still has no product revenues, just collaboration revenues from its research agreement with Merck (MRK). That deal hinted at JANX’s value with its TRACTr technology, as MRK also wants to use it to develop its own T-cell engager drugs. I suspect there could be more collaborations like these because developing safe, novel T-cell engagers precisely is groundbreaking. So, despite JANX’s promising results on its internal drug pipeline (i.e., JANX007 and JANX008), its platform sets JANX apart.
It’s also worth mentioning on April 10, 2024, JANX’s stock price increased 12% due to potential takeover interest from big pharma companies wanting to broaden their portfolios. Naturally, we have few details on these potential M&A offers, and it’s impossible if they will materialize, but I suspect there’s something to them. In my view, it would make sense if JANX’s platform is as effective and versatile for producing new drug candidates as MRK’s agreement suggests. Regardless of the outcome, the attention of big pharma companies highlights the value and potential impact of JANX’s TRACT technology.
Source: Seeking Alpha.
On the other hand, much of JANX’s value today seems related to such M&A speculation. Still, it’s also true that JANX has substantial financial resources. Its latest balance sheet report showed $212.8 million in cash and equivalents and $439.0 million in short-term investments. So, adding these two, JANX has essentially $651.8 million in available short-term liquidity. It also has no financial debt, only $33.2 million in total liabilities, most from operating lease obligations.
JANX’s cash burn is also somewhat moderate compared to its available funds. By adding its CFOs and Net CAPEX, I estimate its latest quarterly cash burn was $15.0 million. This implies a yearly cash burn of $60.0 million, which suggests a long cash runway of about 10.9 years. This means the company has more than enough resources to fund its research for the foreseeable future, even though it’s still mostly in Phase 1.
JANX’s T-cell engagers are part of the broader T-cell therapy market. (Source: GrandViewResearch.)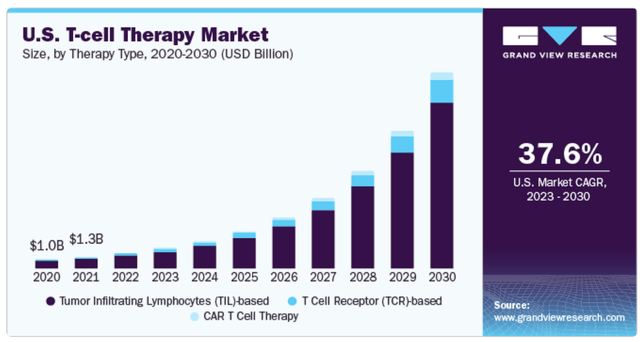
Moreover, its most recent book value stood at $655.8 million, implying a 3.2 P/B. For reference, its sector median P/B multiple is 2.36. So, JANX trades at a slight premium relative to peers. Still, it’s worth considering that its underlying IP appears potentially quite valuable, so I think its valuation is reasonable on balance. Hence, JANX seems a reasonable “buy” for investors who want to bet on this potentially disruptive platform and technology. It also has promising drug candidates in Phase 1 with results that pave the way to further trials in the near term. Since T-cell engagers are part of the broader T-cell therapy market, there’s ample potential for JANX’s IP if they’re successful.
Investment Caveats: Risk Analysis
Nevertheless, I must highlight that the main risk to my “buy” thesis is that JANX’s platform is disappointing. As I previously noted, much of its current valuation is likely due to its speculative attractiveness to other pharma giants. If further clinical trials on JANX007 and JANX008 somehow suggest that the company’s technology isn’t as viable as it appears today, then the stock would likely suffer accordingly.
Source: TradingView.
Plus, it’s important to consider the potential stock price implications if the ongoing M&A rumors are disproven or completely discarded. Yet, as a standalone business, I believe JANX’s ample resources and platform position it well for delivering shareholder value over the long term. After all, JANX’s promising clinical trial results for these drug candidates showed their effectiveness and safety, drawing the attention of big pharma companies and demonstrating the significant role that JANX will play in advancing cancer treatments.
Buy: Conclusion
Overall, JANX has a potentially revolutionary platform with its TRACTr technology. Two promising drug candidates are progressing well along the FDA’s regulatory pathway, and Phase ⅔ is likely to start in the next year or two. In my estimation, JANX’s balance is also quite robust, with a long cash runway of over ten years. Lastly, the recent M&A rumors also add to its potential, as a takeover would probably return significant value to shareholders. So, despite the inherent uncertainty of JANX’s technology, I think its investment equation is ultimately favorable. Hence, I deem JANX a “buy” for investors seeking to gain oncology biotech exposure.
Read the full article here