Introduction
Precigen (NASDAQ:PGEN), a Virginia-based biopharmaceutical company, focuses on advancing gene and cell therapies to improve patient outcomes for unmet medical needs. The company uses proprietary technology platforms to develop products for urgent diseases in immuno-oncology, autoimmune disorders, and infectious diseases. Subsidiaries include Precigen ActoBio, creating microbe-based therapeutics in Belgium, and Precigen Exemplar, developing MiniSwine research models and regenerative medicine applications in Iowa.
Precigen pipeline (Precigen)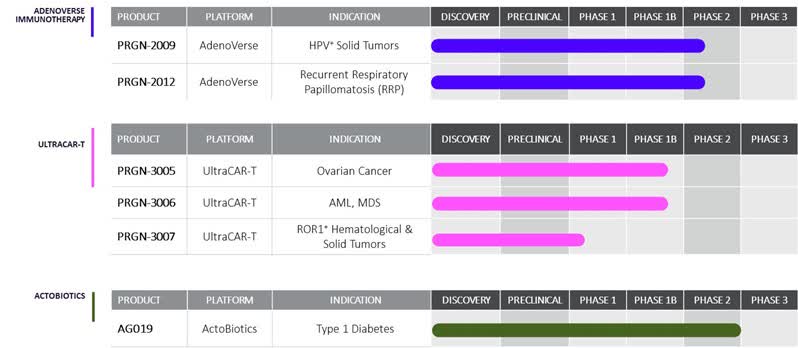
This article examines Precigen’s latest financial report and clinical advancements while delving into a comprehensive analysis of their primary drug candidate, PRGN-2012.
Q1 2023 Financials
In Q1 2023, Precigen reported total revenues of $1.85 million, significantly down from $5.51 million in Q1 2022. Operating expenses were lower at $25.33 million, compared to $27.67 million the previous year, with R&D costs rising slightly. The company recorded an operating loss of $23.48 million, a slight increase from the $22.15 million loss in 2022. Total other income saw an increase to $0.69 million from a net loss of $1.80 million in 2022, primarily due to lower interest expenses and higher interest income. The net loss from continuing operations was $22.73 million, down from $23.90 million.
Per Seeking Alpha, Precigen has a market capitalization of $329.57 million, total debt amounting to $21.69 million, and cash reserves of $104.09 million. The company’s enterprise value stands at $247.17 million.
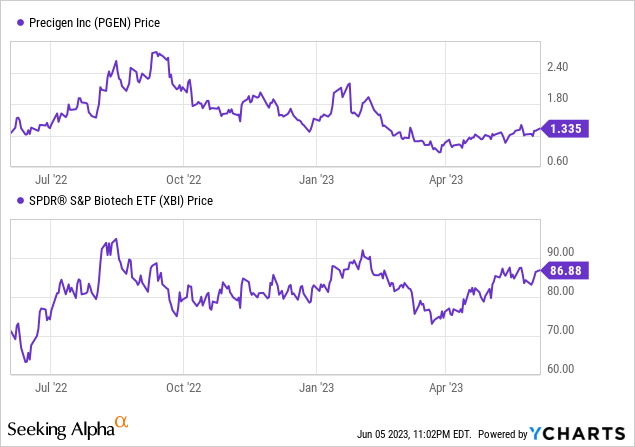
Q1 2023 Earnings Call Review
During the recent earnings call, Precigen’s management discussed significant progress with several ongoing clinical programs and expressed excitement about the potential of their therapeutic candidates.
-
PRGN-2012: It is being used to treat recurrent respiratory petaloma disease, a rare condition for which there are currently no therapies. Management reported favorable safety data and a complete response rate of 50% in severe patients following 12 months of use. They also mentioned productive discussions with the FDA regarding the therapy.
-
PRGN-2009: The drug is currently in clinical trials for treating HPV-related cancers, such as cervical and head and neck cancers. Management expressed anticipation for forthcoming data from the trial, particularly regarding efficacy in patients who have failed all other therapies.
-
UltraCAR-T Platform: Several clinical trials are ongoing with different UltraCAR-T candidates (PRGN-3006, PRGN-3005, PRGN-3007). These therapies involve modifying patient T cells to target specific cancer cells, a process that the company can achieve overnight. Management highlighted preliminary efficacy data and promising safety profiles.
-
Rights regained for CD19 and BCMA: The company regained rights to these validated targets, which are used in CAR-T therapies. Precigen expects to be Phase I ready for CD19 by the end of the year, with BCMA to follow. They believe their platform, combined with these targets, can offer cost-effective, innovative treatments to patients.
-
Rights regained for IL-12: Precigen has also regained rights to IL-12. They see potential in combining IL-12 with their current molecules for future therapies, highlighting the importance of IL-12 in enhancing immune responses.
Let’s take a closer look at Precigen’s most advanced immunotherapy, PRGN-2012.
PRGN-2012 Immunotherapy Shows Promise for RRP
The Phase 1 clinical trial (NCT04724980) assessed PRGN-2012, a novel immunotherapy intervention, for treating Recurrent Respiratory Papillomatosis (RRP). The trial involved adult patients with aggressive RRP, who had three or more surgeries within the preceding 12 months. Out of the 15 participants enrolled in the study, three received a lower dose (Dose Level 1), while 12 were given a higher dose (Dose Level 2) of PRGN-2012. It was administered four times via subcutaneous injection.
The patient group predominantly consisted of males (10 out of 15), with a median age of 51 years. These patients had an average of 6.2 surgeries in the last year before the trial and had been diagnosed with RRP for an average of 15 years.
The drug demonstrated acceptable safety, with no severe treatment-related adverse events (TRAEs) reported. All participants experienced a mild injection site reaction, while other TRAEs were rare and diminished in frequency over time.
In terms of clinical efficacy, the results seem promising but should be interpreted with caution given the limitations of the data. One primary concern is the small sample size. While 50% (6 out of 12) of patients at Dose Level 2 showed a complete response, meaning they didn’t require surgery during the 12-month follow-up, these results need to be validated in larger patient cohorts. Moreover, long-term follow-up data is needed to assess the durability of the response. It’s also crucial to investigate potential side effects that may only become evident over a longer period.
RRP, mainly caused by HPV 6 and HPV 11, leads to benign tumor growth in the respiratory tract, often necessitating repeat surgeries. Given the recurrent nature of RRP and the significant burden of repeated surgeries, PRGN-2012 could potentially fit well into the treatment paradigm if its efficacy and safety are confirmed in larger, longer-term trials. PRGN-2012’s ability to stimulate HPV 6/11-specific T-cell response suggests that it could provide a durable response and reduce the recurrence of papillomas.
As for the market, while RRP is a rare disease, the need for an effective, less invasive treatment could provide a niche yet impactful market for PRGN-2012. Presently, the primary treatment for RRP is surgery, which doesn’t prevent recurrence and can lead to complications over time. An effective immunotherapy could transform the treatment landscape for RRP, improving patient outcomes and potentially reducing healthcare costs associated with repeated surgeries. It’s important to note, however, that market success would also depend on factors such as pricing, reimbursement policies, and the competitive landscape.
My Analysis & Recommendation
Given the prevailing scenario, Precigen appears to be in the throes of an innovation cycle in the bio-pharmaceutical landscape. The company’s financial outlook may initially seem less than optimistic, with decreasing revenues and sustained operating losses. However, the broader picture presents a more nuanced narrative that investors must pay attention to.
Significant strides are being made in several of the company’s clinical programs, notably PRGN-2012 and PRGN-2009. While still in the early stages, the positive safety data, promising efficacy indicators, and successful discussions with the FDA regarding PRGN-2012 show potential for growth and innovation in a currently unmet medical need. This adds a layer of intrinsic value to Precigen’s position.
Similarly, the ongoing clinical trials for PRGN-2009 may unlock new avenues in the treatment of HPV-related cancers, particularly for patients who have exhausted other therapeutic options. This also underscores Precigen’s potential for innovative disruption in the immuno-oncology field.
Moreover, the reacquisition of rights for CD19, BCMA, and IL-12 could catalyze the development of more effective CAR-T therapies. By leveraging their proprietary UltraCAR-T platform, Precigen might be uniquely positioned to transform treatment paradigms, not only improving patient outcomes but also demonstrating cost-effective models in an increasingly cost-conscious healthcare environment.
However, uncertainties remain. The success of the company’s trials, regulatory approval, market acceptance, and successful commercialization of the therapies will be crucial to its financial recovery. Investors should also consider the company’s cash burn rate relative to its cash reserves, the ability to raise additional funds if needed, and the dynamics of the biotech market in the face of competitive forces.
In conclusion, although Precigen may seem to be in choppy waters financially, its underlying clinical potential could outweigh the immediate drawbacks. In view of the high-risk, high-reward nature of the biotech sector, and considering Precigen’s forward-looking innovations in gene and cell therapy, my investment recommendation for Precigen is a “Hold”. This recommendation acknowledges the need for investors to stay attuned to further developments, while acknowledging the firm’s promise in the face of its prevailing challenges. As such, investors with a high-risk tolerance and a long-term investment horizon may see a potential upside in Precigen’s clinical advancements.
Read the full article here